How a $33 billion medical device giant built a modular surgical robot to challenge Intuitive Surgical’s 25-year monopoly, and why an NVIDIA supercomputing partnership may define its next chapter.
On February 17, 2026, Dr. Jihad Kaouk, professor and chair of the Glickman Urologic Institute at Cleveland Clinic, performed the first commercial surgery in the United States using the Hugo RAS system: a robotic-assisted prostatectomy (Medtronic, 2026). The patient was discharged the very next day. After decades of a single company dominating the operating room, a $130 billion healthcare giant has entered surgical robotics with a machine engineered to break that dominance. The question is not whether Medtronic can build a competitive robot. It already has. The question is whether hospitals, surgeons, and health systems are willing to change.
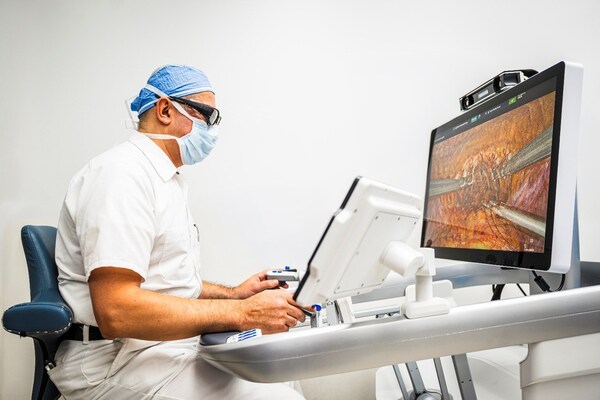
Section 1: The Company Behind the Machine
Medtronic plc traces its origins to a garage in Minneapolis, Minnesota, where Earl Bakken and Palmer Hermundslie founded the company in 1949 (Medtronic, 2025). From those modest beginnings, Bakken’s team invented the first wearable external cardiac pacemaker in 1957, a device that fundamentally redefined the boundary between life and death for patients with heart rhythm disorders. That single invention established a blueprint the company has followed ever since: identify an unmet clinical need, engineer a novel solution, and distribute it at scale.
Today, Medtronic is the world’s largest medical device company by revenue. The company employs more than 95,000 people across more than 150 countries and develops technologies that treat 70 distinct health conditions (Medtronic, 2025). Its stated mission, unchanged in its core intent since Bakken articulated it, is “to alleviate pain, restore health, and extend life.” Annual revenue stands at approximately $33 billion, a figure that reflects the company’s breadth across cardiac rhythm management, spine surgery, diabetes care, neurology, and surgical technologies.
Two acquisitions shaped the modern Medtronic in critical ways. In 2015, the company acquired Covidien for $49.9 billion, a transaction that instantly made Medtronic the world’s largest medical device company and provided a dominant position in surgical instruments, energy devices, and laparoscopic tools (Medtronic, 2025). In 2018, Medtronic acquired Mazor Robotics for $1.64 billion, purchasing the Israeli company’s robotics guidance platform for spine surgery and, more importantly, the institutional knowledge to develop and commercialize robotic surgical systems at scale. Those two acquisitions, spanning the full spectrum from laparoscopic instruments to surgical robotics, set the table for what came next.
Medtronic’s surgical business now spans all three operative modalities: open surgery, laparoscopic surgery, and robotic-assisted surgery. No other company in the world can claim the same coverage, a fact that Medtronic’s leadership has cited repeatedly as a foundational competitive advantage (Medtronic, 2026).
Image credit: Medtronic plc
Section 2: What Hugo RAS Was Built to Solve
To understand the Hugo RAS system, one must first understand the problem it was designed to address. Intuitive Surgical has dominated robotic-assisted surgery for approximately 25 years, building an installed base of roughly 9,500 da Vinci surgical systems worldwide and a market capitalization that reached approximately $170 billion in early 2026 (Intuitive Surgical, cited in Surgical Science, 2025; companiesmarketcap.com, 2026). The da Vinci platform generates recurring revenue from instruments and service contracts, creating powerful financial incentives for hospitals to remain within the Intuitive ecosystem once they have invested in the initial system.
The result is a market with a significant access problem. Da Vinci systems carry price tags ranging from approximately $1.5 million to $2.5 million for newer configurations, and each system typically requires a dedicated operating room and a substantial training investment before surgeons reach proficiency (R2 Surgical, 2025). Hospitals in smaller markets, community health settings, and developing countries have largely been priced out of robotic surgery entirely. Even in large academic medical centers, robotic capacity frequently constrains the number of minimally invasive procedures that can be performed.
Hugo’s architecture was designed with these constraints in mind. The system uses a modular, multi-arm design in which individual robotic arms can be repositioned and theoretically shared across different operating rooms within a hospital, improving capital utilization relative to a fixed single-room installation. The surgeon console is open rather than enclosed, a deliberate departure from the da Vinci cockpit experience: the open design is intended to improve the surgeon’s situational awareness and facilitate real-time communication with the scrub team and anesthesia staff during procedures (Medtronic, 2026).
Hugo also integrates with Medtronic’s Touch Surgery ecosystem, a software platform that provides AI-powered surgical video management, performance analytics, and remote tele-proctoring capabilities. Tele-proctoring is particularly significant for adoption in lower-volume centers, where a surgeon learning a new robotic platform can receive real-time guidance from an experienced colleague located at a different institution, without requiring that expert to be physically present in the room.
Rajit Kamal, vice president and general manager of Robotic Surgical Technologies at Medtronic, described the company’s positioning directly: “The future of surgery isn’t about robotics as a separate modality, it’s about technology that enables surgical teams to deliver the best possible care for every patient. Medtronic is uniquely positioned to do that as the only company in the world that can partner with surgeons and hospitals across open, laparoscopic, and robotic-assisted surgery” (Medtronic, 2026).
Section 3: The NVIDIA Partnership and the GTC 2026 Announcement
At NVIDIA’s GTC 2026 conference in San Jose in March 2026, the company announced that Medtronic is among the organizations currently evaluating NVIDIA’s IGX Thor platform for surgical robotics applications (NVIDIA, 2026). The announcement placed Medtronic alongside a range of industrial partners, including Caterpillar, Hitachi Rail, and KION, as well as other healthcare companies including Johnson and Johnson MedTech and KARL STORZ.
IGX Thor is an industrial-grade edge computing platform built for real-time physical AI at the edge. The platform delivers high-speed sensor processing, enterprise-grade reliability, and functional safety certifications, characteristics that are prerequisites for deployment in safety-critical environments such as operating rooms. Its core technical capability is the processing and orchestration of multimodal sensor data, including video feeds, imaging data, and device telemetry, through low-latency AI pipelines that can support real-time inference during active procedures (NVIDIA, 2026).
The platform is built on NVIDIA’s Holoscan software framework, which is specifically designed for next-generation software-defined medical devices and intelligent operating rooms. Holoscan provides the middleware layer between the raw sensor hardware and AI inference models, enabling developers to build applications that process multiple data streams simultaneously without introducing the latency that would make real-time surgical guidance impractical.
The potential applications in a robotic surgery context are significant. Real-time tissue recognition could enable a system to identify anatomical structures and flag them to the surgeon before an instrument makes contact. Instrument tracking could provide automated safety boundaries around critical structures. Anomaly detection could alert the surgical team to unexpected findings during the procedure itself, before the case concludes. None of these capabilities are currently deployed in the Medtronic-NVIDIA integration: as of March 2026, the relationship remains in an evaluation phase (NVIDIA, 2026). However, the direction of the partnership is clear.
NVIDIA also unveiled at GTC 2026 a suite of open healthcare robotics resources, including Open-H, a dataset comprising 776 hours of surgical video drawn from 11 robotic system embodiments across 4 surgical indications, developed with 35 collaborators (NVIDIA, 2026). Companion tools including the Cosmos-H synthetic data generation model and the GR00T-H vision-language-action model are designed to accelerate the training of AI systems for surgical environments. These resources are available to the broader developer ecosystem, including Medtronic and its engineering partners.
Section 4: The Flagship Product
The Hugo RAS system received FDA clearance on December 3, 2025, for minimally invasive urologic surgical procedures including prostatectomy, nephrectomy, and cystectomy (Medtronic, 2025). These three procedure categories collectively represent approximately 230,000 surgeries per year in the United States, a substantial addressable market for an initial indication (Medtronic, 2025). The clearance followed completion of the Expand URO investigational device exemption clinical study, which Medtronic describes as the largest ever completed for multi-port robotic-assisted urological surgery in the United States (Medtronic, 2025). The study demonstrated that the Hugo RAS system met primary safety and effectiveness endpoints, with outcomes consistent with published literature for comparable robotic and laparoscopic procedures.
The first commercial case in the United States was performed at Cleveland Clinic on February 17, 2026. Dr. Kaouk’s team completed the robotic-assisted prostatectomy without complication, and the patient was discharged the following day (Medtronic, 2026). Duke University Hospital and Atrium Health Wake Forest Baptist were also among the first U.S. institutions to receive Hugo installations, representing a deliberate strategy of partnering with nationally recognized academic medical centers to build credibility and generate clinical publications before broader commercial rollout.
Internationally, Hugo has a significantly more established track record. The system is available in more than 35 countries and has been used in tens of thousands of urologic, gynecologic, and general surgery procedures across more than five continents (Medtronic, 2025; Medtronic, 2026). Outside the United States, Hugo has already expanded into gynecologic and general surgery indications. In the U.S. market, those additional clearances are pending, with Medtronic signaling planned expansion to those specialties as a near-term priority following the initial urology launch.
Pricing for the Hugo RAS system has not been publicly disclosed by Medtronic. Industry analysts and procurement specialists have suggested that the company is positioning Hugo competitively with or below da Vinci system pricing, consistent with the company’s stated goal of expanding access to robotic surgery in settings where cost has historically been a barrier.
Dr. James Porter, chief medical officer for Robotic Surgical Technologies at Medtronic, characterized the product’s positioning at the time of FDA clearance: “The Hugo RAS system represents a new and exciting approach to robotic-assisted surgery. We’re excited for surgical teams in the U.S. to experience the differentiated technology and partnership from Medtronic, which supports them at every stage of their robotic surgical journey” (Medtronic, 2025).
Section 5: Market and Economic Context
The surgical robotics market has entered a period of rapid expansion. According to MarketsandMarkets, the global surgical robots market was valued at approximately $13.69 billion in 2025 and is projected to reach $27.14 billion by 2030, representing a compound annual growth rate of 14.7% (MarketsandMarkets, 2025). This growth is driven by aging populations in high-income countries, increasing preference for minimally invasive procedures, and the gradual diffusion of robotic platforms into community hospital settings beyond the large academic medical centers that were early adopters.
Within this market, Intuitive Surgical occupies a commanding position, but its share is under pressure for the first time in years. The da Vinci 5, launched in 2024, represents the most significant hardware upgrade in the platform’s history, incorporating force feedback and enhanced ergonomics. However, the broader competitive landscape has shifted materially: Medtronic, Johnson and Johnson’s Ottava platform, and CMR Surgical’s Versius system are all now in various stages of U.S. commercialization or regulatory progression.
The urology robotic surgery segment alone was valued at approximately $2 billion in 2022 and is expected to grow at a CAGR of approximately 8% through 2033, according to data cited by Zacks Equity Research (Yahoo Finance, 2025). Robotic assistance already accounts for roughly 80% of urologic abdominal surgeries in the United States, making urology simultaneously the most mature robotic surgery market and the segment with the most established reimbursement pathways for new entrants (Medtronic, 2026).
| Metric | Details |
|---|---|
| Global surgical robotics market size (2025) | ~$13.69 billion |
| Projected market size (2030) | ~$27.14 billion |
| Projected CAGR (2025 to 2030) | 14.7% |
| Intuitive Surgical market cap (March 2026) | ~$170 billion |
| Intuitive installed base (da Vinci) | ~9,500 systems |
| Hugo RAS countries available | 35+ |
| Hugo procedures globally (as of early 2026) | Tens of thousands |
| U.S. urologic procedures (annual, target indications) | ~230,000 |
| Medtronic total annual revenue | ~$33 billion |
| Medtronic employees | 95,000+ across 150+ countries |
Section 6: Concerns and Counterarguments
The case for Hugo is compelling on paper. The execution risks are equally substantial, and any serious analysis must engage with them directly.
The switching cost problem. Intuitive Surgical’s 25-year head start translates into more than just installed systems. It translates into surgical training programs built around da Vinci, residents who have never operated any other robotic platform, and hospital procurement committees that have learned to manage Intuitive contracts over decades. The da Vinci 5, launched in 2024, added force feedback and ergonomic improvements that represent a genuine step forward in the technology, reinforcing the case for hospitals to deepen their existing Intuitive relationship rather than invest in a second platform (R2 Surgical, 2025).
Limited initial indication. Hugo’s FDA clearance covers urologic procedures only. Gynecology and general surgery, which represent a significant share of the total robotic surgery market, require separate regulatory clearances that Medtronic has not yet obtained in the United States. Competing platforms, including the da Vinci system, already hold clearances across a broader range of specialties, which limits Hugo’s immediate commercial opportunity and its appeal to surgical departments looking for a single platform solution.
Capital budget constraints. Hospital capital budgets remain under significant pressure following a period of elevated labor costs, supply chain disruptions, and post-pandemic financial recovery. Persuading a hospital’s capital allocation committee to add a second robotic platform to an operating room suite that already has da Vinci capacity is a difficult proposition, even when the incremental cost is competitive. Medtronic must demonstrate not just clinical equivalence but operational and financial ROI that is sufficiently compelling to justify the investment.
Mixed execution history. Medtronic’s robotics ambitions have not always been realized on the originally projected timeline. The company’s broader strategic pivot toward surgical robotics has proceeded more slowly than some analysts anticipated following the Mazor and Covidien acquisitions, and Hugo’s commercial launch in the United States came later than some initial projections suggested (Yahoo Finance, 2025). While the FDA clearance and successful first cases are real milestones, the commercial scaling phase carries its own execution risks.
The NVIDIA integration is early stage. As of March 2026, Medtronic is described as “evaluating” IGX Thor, which is meaningfully different from a deployed clinical capability (NVIDIA, 2026). The path from evaluation to a regulatory-cleared, clinically validated, real-time AI inference capability in an active surgical system is measured in years, not quarters. The partnership signals strategic intent; it does not yet represent a product feature.
Surgeon training timelines. Robotic surgery platforms require surgeons to complete proctored training cases before operating independently. Hospitals that commit to Hugo must invest in that training process across multiple surgeons over an extended period before the platform’s utilization can scale to a level that justifies the capital expenditure. That timeline represents both a barrier to adoption and a period of vulnerability during which competitive programs at rival institutions may capture procedural volume.
Conclusion
Medtronic is not attempting to defeat Intuitive Surgical by building a superior robot. It is attempting to reach surgical settings that Intuitive has not fully penetrated, by offering a modular, shareable system with lower friction for adoption and a software ecosystem designed to support surgeons throughout their learning curve rather than simply at the moment of purchase. The NVIDIA IGX Thor partnership points toward the logical endpoint of that strategy: a surgical platform that combines physical dexterity with real-time AI inference, enabling a level of intraoperative decision support that current systems do not provide.
Whether Hugo can translate its international experience, its modular architecture, and its parent company’s commercial reach into a durable position in the U.S. market remains the central question. The first commercial case at Cleveland Clinic demonstrated that the technology works and that patients can go home the next day. Demonstrating that at scale, across hundreds of hospitals and thousands of surgeons, is the harder problem. Medtronic has the capital, the distribution infrastructure, and the regulatory expertise to attempt it. The surgical robotics market, now projected to exceed $27 billion by 2030, is large enough to support multiple major platforms (MarketsandMarkets, 2025). The open question is whether hospitals will agree.
References
- Medtronic. (2026, February 17). Medtronic announces first surgery with Hugo robotic-assisted surgery system in the U.S. performed at Cleveland Clinic. https://news.medtronic.com/2026-02-17-Medtronic-announces-first-surgery-with-Hugo-TM-robotic-assisted-surgery-system-in-the-U-S-performed-at-Cleveland-Clinic
- Medtronic. (2025, December 3). Medtronic announces FDA clearance of Hugo robotic-assisted surgery system for urologic surgical procedures. PR Newswire. https://www.prnewswire.com/news-releases/medtronic-announces-fda-clearance-of-hugo-robotic-assisted-surgery-system-for-urologic-surgical-procedures-302632328.html
- Medtronic. (2025). Hugo robotic-assisted surgery. https://www.medtronic.com/en-us/healthcare-professionals/specialties/surgical-robotics/hugo-robotic-assisted-surgery.html
- Medtronic. (2025). Company history. https://www.medtronic.com/en-us/our-company/history.html
- NVIDIA. (2026, March 16). NVIDIA and global robotics leaders take physical AI to the real world. NVIDIA Investor Relations. https://investor.nvidia.com/news/press-release-details/2026/NVIDIA-and-Global-Robotics-Leaders-Take-Physical-AI-to-the-Real-World/default.aspx
- NVIDIA. (2026, March 20). NVIDIA GTC 2026: Live updates on what’s next in AI. NVIDIA Blog. https://blogs.nvidia.com/blog/gtc-2026-news/
- Zacks Equity Research. (2025, December 5). MDT stock set to benefit from the FDA clearance of Hugo RAS system. Yahoo Finance. https://finance.yahoo.com/news/mdt-stock-set-benefit-fda-134800163.html
- MarketsandMarkets. (2025, December 9). Surgical robots market worth $27.14 billion by 2030. Yahoo Finance. https://finance.yahoo.com/news/surgical-robots-market-worth-27-153000100.html
- Surgical Science. (2025, January 15). Surgical Science is transitioning to a fully subscription-based revenue model with Intuitive and all da Vinci 5 systems. https://surgicalscience.com/mfn_news/surgical-science-is-transitioning-to-a-fully-subscription-based-revenue-model-with-intuitive-and-all-da-vinci-5-systems-will-be-equipped-with-surgical-science-simulation-software/
- R2 Surgical. (2025). How much is a surgical robot? (2025 edition). https://r2surgical.com/blogs/x-and-xi-robots/how-much-is-a-surgical-robot-2025-edition
- companiesmarketcap.com. (2026). Intuitive Surgical market capitalization. https://companiesmarketcap.com/intuitive-surgical/marketcap/
