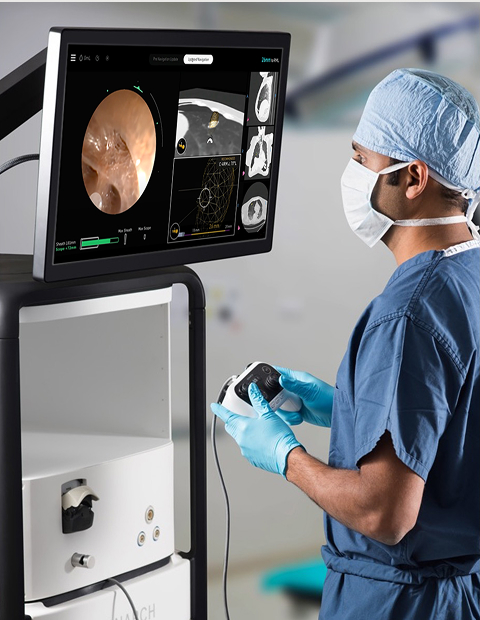
How Johnson & Johnson MedTech is using a flexible robot, artificial intelligence, and NVIDIA simulation to transform the diagnosis of lung cancer and reshape the competitive landscape of surgical robotics.
The Monarch Platform. Image credit: Johnson & Johnson MedTech
Lung cancer is the deadliest cancer on earth. It kills more people each year than breast, colon, and prostate cancers combined. Yet early detection, the single most powerful tool for improving survival, has long been stymied by a basic anatomical problem: the tumors form deep inside airways that human hands cannot safely reach. Now, a flexible robot roughly the diameter of a pencil is navigating those airways for surgeons, finding lesions in the lung periphery and enabling biopsies that were once impossible without invasive surgery. That robot is the Monarch Platform, developed by Auris Health and owned by Johnson & Johnson MedTech. It is one of the most consequential bets in the medical device industry today.
Section 1: The Company Behind It
Johnson & Johnson is the world’s largest healthcare company, and its MedTech segment is a major driver of that scale. In 2025, J&J MedTech recorded $33.79 billion in sales, representing 6.1% year-over-year growth, with cardiovascular devices leading the charge at $8.93 billion in sales and nearly 16% growth (MedTech Dive, 2026). MedTech contributes approximately 36% of J&J’s overall revenue (Yahoo Finance, 2026).
The Monarch Platform entered the J&J portfolio through one of the more significant acquisitions in recent medtech history. In 2019, Johnson & Johnson acquired Auris Health for $3.4 billion, bringing the Monarch robotic bronchoscopy system into its MedTech fold (MedTech Dive, 2019). Auris Health was founded in 2007 by Frederic Moll, a name well known in surgical robotics: Moll is also a co-founder of Intuitive Surgical, the company behind the da Vinci system. His involvement lent the Auris platform significant technical credibility from its earliest days.
J&J’s strategy in MedTech is sharpening around higher-growth segments. The company has announced plans to spin off its orthopedics business by mid-2027, allowing it to concentrate resources on cardiovascular devices and surgical robotics (MedTech Dive, 2026). According to Yahoo Finance (2026), the company has been systematically repositioning its MedTech assets: about half of those assets are now in higher-growth markets, up from just 20% in 2018, with the orthopedic spin-off expected to push that figure above 70%. The Monarch Platform sits directly in that higher-growth zone.
Section 2: What the Monarch Platform Solves
The clinical problem the Monarch Platform addresses is both simple to state and difficult to solve. Lung cancer kills more people globally than breast, colon, and prostate cancers combined. Early detection is critical to survival, but lung nodules are often tiny, located deep in the peripheral airways, and practically inaccessible to conventional diagnostic tools. Traditional bronchoscopy instruments have limited reach; they cannot navigate safely into the outer regions of the lung where many early-stage tumors form. CT-guided transthoracic needle biopsy, the alternative, is more invasive and carries meaningful risks including pneumothorax.
The Monarch Platform is the first robotic-assisted bronchoscopy system to reach the market (JNJ.com, 2025). It uses a flexible robotic endoscope in a novel scope-in-sheath architecture, allowing the physician to navigate through the patient’s natural airways using a controller that operates on principles similar to a video game interface. The system provides continuous visualization through a dedicated camera and a separate instrument channel, so the physician maintains sight of the operative field throughout the biopsy process.
According to J&J MedTech (2025), nearly half of early-stage lung lesions are located in the periphery of the lung, regions that are difficult or impossible to reach with traditional bronchoscopy. The platform is designed to address precisely that gap. Early clinical results from the Benefit study, presented at the American Thoracic Society conference, showed that the Monarch system successfully localized lung nodules in 92% of patients, with no significant adverse events reported (MedTech Dive, 2019). Research published in the Journal of Clinical Medicine (2026) found comparable diagnostic yields between Monarch and competing platforms in real-world clinical settings.
The platform’s clinical reach now extends beyond the lung. J&J received FDA clearance for a urology indication in 2022, enabling the Monarch system to be used in kidney stone procedures. That urology platform is slated for its U.S. commercial launch in 2026 (MedTech Dive, 2025). The system is indicated for endoscopic visualization and access of organs and canals in the urinary tract, providing precise instrument control and allowing a single operator to manage multiple tasks simultaneously during kidney stone procedures (JNJ.com, 2025).
The Monarch Quest software update, cleared by the FDA in March 2025, represents the most significant recent upgrade to the bronchoscopy platform. Powered by a full-stack NVIDIA accelerated computing platform based on NVIDIA RTX, Monarch Quest increases real-time computational power by 260% and integrates AI-powered navigation algorithms. It also includes a verified interface with the GE HealthCare OEC 3D mobile CBCT imaging system, combining enhanced navigation with intraprocedural 3D imaging to improve accuracy in targeting nodules. The update is part of J&J’s Polyphonic digital ecosystem, which encompasses preoperative planning, intraoperative navigation, and post-operative case data analysis (JNJ.com, 2025).
Section 3: The NVIDIA Partnership
The most forward-looking chapter of the Monarch Platform’s development is its deepening integration with artificial intelligence, particularly through a partnership with NVIDIA announced in October 2025 and highlighted further at GTC 2026 in March of that year. J&J MedTech is using NVIDIA Isaac for Healthcare, a developer framework for medical robotics, to simulate how the Monarch system performs inside virtual operating rooms and inside virtual patient anatomy (MedTech Dive, 2025).
The technical architecture of this partnership has two main components. First, NVIDIA Isaac Sim, built on NVIDIA Omniverse libraries, creates high-fidelity digital twins of the operating room environment: towers, carts, fluid management systems, instruments, and the physical layout of the surgical suite. These simulations allow J&J’s R&D teams to test new features and model how the system will behave in real-world conditions before any physical prototype is built. Second, NVIDIA Cosmos world foundation models generate synthetic training data for the robot’s AI systems, providing highly accurate, physics-based data that reflects the behavior of tissues, fluids, and instruments in realistic clinical scenarios (JNJ.com, 2025; Surgical Robotics Technology, 2025).
The practical implication is significant. Rather than relying solely on cadaver studies, animal models, or limited human trials to train AI algorithms, J&J can now expose the Monarch’s AI to millions of simulated clinical scenarios before the system ever touches a patient. For the urology platform specifically, simulations include modeled kidney stone procedures across diverse patient anatomies and procedural setups, creating virtual environments for both R&D development and clinical team training.
Neda Cvijetic, Senior Vice President and Global Head of Robotics and Digital R&D at Johnson & Johnson MedTech, described the ambition directly: “Simulation is the next frontier in surgical robotics. With AI-driven simulation, we can create high-fidelity digital twins that adhere to the laws of physics, such that the simulation accurately anticipates the real world and ultimately unlocks physical AI capabilities” (Surgical Robotics Technology, 2025).
At GTC 2026, NVIDIA highlighted its expanding global robotics ecosystem partnerships, bringing together industrial and surgical robot companies to develop and deploy physical AI at scale (NVIDIA Investor Relations, 2026). J&J’s Monarch work sits within that broader strategy: using simulation and foundation models to compress development timelines and reduce the dependency on physical testing.
Section 4: The Flagship Product as a Business
From a commercial perspective, the Monarch Platform is a classic capital-intensive medical device business. It is a B2B product sold directly to hospitals, academic medical centers, and surgical centers. J&J has not publicly disclosed the pricing of the Monarch system, but comparable robotic surgical platforms, including Intuitive Surgical’s da Vinci systems, typically carry capital costs in the range of $1.5 million to $3 million or more per installation. Beyond the initial capital sale, the revenue model depends on per-procedure consumables and software subscription services, creating recurring revenue streams tied to procedure volume.
The addressable market for robotic-assisted bronchoscopy is a small but fast-growing segment of the lung biopsy market in the United States. As of mid-2023, analysts estimated that robotic platforms had penetrated only around 4.7% of the bronchoscopy market, leaving substantial room for expansion (MedTech Dive, 2023). The broader surgical robotics market was valued at approximately $8.28 billion in 2025 and is projected to reach $16.40 billion by 2032, reflecting a compound annual growth rate of roughly 10% (MMR Statistics, 2026).
The principal competitors in the robotic bronchoscopy space are Intuitive Surgical’s Ion Endoluminal System and Noah Medical’s Galaxy System. Ion received FDA clearance in 2019, one year after Monarch, and has built a substantial installed base. Noah Medical’s Galaxy differentiates itself with a single-use disposable bronchoscope and integrated tomosynthesis technology for real-time navigation updates (Noah Medical).
Section 5: Surgical Robotics Competitive Landscape
| Company | System | Primary Use | FDA Cleared | Approx. Installed Base / Status |
|---|---|---|---|---|
| J&J MedTech | Monarch Platform | Bronchoscopy, Urology | Yes (2018 bronchoscopy; 2022 urology) | Growing; launched 2018; urology commercial launch 2026 |
| Intuitive Surgical | da Vinci / Ion | General surgery / Bronchoscopy | Yes | 9,500+ da Vinci systems globally; Ion installed base growing rapidly |
| Medtronic | Hugo RAS | Urology, General Surgery | Yes (December 2025) | Early U.S. commercial launch underway; first case at Cleveland Clinic, February 2026 |
| CMR Surgical | Versius / Versius Plus | General Surgery | Yes (October 2024 / December 2025) | 40,000+ procedures globally outside U.S.; U.S. commercial launch 2026 |
| Noah Medical | Galaxy System | Bronchoscopy | Yes (2023) | Growing; single-use bronchoscope differentiation |
Sources: Medtronic News (2026); CMR Surgical (2025); Noah Medical; MedTech Dive (2023).
The table above illustrates the increasingly crowded terrain that J&J’s Monarch must navigate. Intuitive Surgical holds an unmatched installed base and brand recognition. Medtronic and CMR Surgical are arriving in the U.S. general surgery robotics market with fresh clearances in late 2025 and 2026. Noah Medical is pressing Monarch directly in the bronchoscopy niche with a compelling disposable-scope design. For Monarch, the differentiation case must rest on its AI capabilities, its urology expansion, and the depth of its clinical integration through the Polyphonic ecosystem.
Section 6: Concerns and Risk Factors
The Monarch Platform carries a set of substantive concerns that any rigorous assessment must address.
Competitive pressure in bronchoscopy. Intuitive Surgical’s Ion system is a direct and formidable rival. A propensity score-matched retrospective study published in the Journal of Bronchology and Interventional Pulmonology in 2025 found that Ion demonstrated a higher diagnostic yield (84.2% vs. 71.0%), shorter robot procedure time, and lower radiation exposure compared to Monarch (PubMed, 2025). A separate study from the Journal of Clinical Medicine found diagnostic yields to be comparable between the two platforms (Ion 75%, Monarch 72%) when advanced imaging tools were not used in either arm (Journal of Clinical Medicine, 2026). The clinical evidence is mixed, and Monarch must continuously improve to stay competitive.
Urology platform still pre-commercial. Despite receiving FDA clearance for the urology indication in 2022, the Monarch urology platform is still in the pre-commercial phase in the United States as of early 2026. Commercial readiness has taken four years to materialize, and execution of that launch will be closely watched.
Niche market constraints. Robotic bronchoscopy is a fast-growing segment, but it remains a niche compared to general surgical robotics. Market penetration remains below 5% of bronchoscopy procedures, limiting near-term revenue potential relative to platforms with broader indications.
AI simulation: promising but unproven at scale. The NVIDIA Isaac and Cosmos partnership is scientifically compelling, but AI trained on synthetic simulation data must still demonstrate its value in real clinical outcomes. Translating physics-based simulations into improved patient results at scale is a meaningful validation hurdle.
OTTAVA delay and broader robotics strategy risk. J&J’s ambitions in surgical robotics extend to the OTTAVA general surgery platform, which targets the massive soft-tissue surgery market dominated by Intuitive Surgical’s da Vinci. OTTAVA submitted a De Novo classification request to the FDA in January 2026, meaning it remains without U.S. marketing authorization as of this writing (JNJ.com, 2026). The prolonged development timeline has raised questions about J&J’s ability to compete in general surgery robotics, and any further delays would weigh on investor confidence in the broader robotics portfolio.
Regulatory risk. Each new clinical indication for the Monarch Platform requires separate FDA clearance. Expanding into new anatomical territories, new disease states, or new procedure types involves regulatory timelines that are inherently unpredictable and can delay commercial plans by years.
Hospital budget pressure. Capital-intensive robotic systems face persistent headwinds from hospital procurement teams managing tight budgets. A platform costing $1.5 million to $3 million competes not only against other robots but against every other capital priority in a health system’s annual budget cycle. Demonstrating clear return on investment and procedure-level economics is essential for widespread adoption.
Conclusion
The Monarch Platform is not a finished product. It is a strategic bet: a declaration that the future of diagnosis and minimally invasive surgery belongs to AI-guided robots that can go where human hands cannot. Johnson & Johnson MedTech has invested $3.4 billion in the acquisition, years of regulatory work across two clinical domains, and a deepening collaboration with NVIDIA to build the simulation infrastructure that will train the next generation of the platform’s AI. With the urology platform approaching commercial launch, Monarch Quest bringing AI-powered navigation to bronchoscopy, and the NVIDIA partnership accelerating development cycles, J&J is positioning Monarch as the foundation of a multi-indication robotic portfolio.
The obstacles are real: a stronger rival in Intuitive Surgical, a crowded competitive field, unproven simulation-to-outcome translation, and a hospital market under financial strain. But if J&J executes on its roadmap, and if the AI navigation advances deliver measurable improvements in diagnostic yield and patient outcomes, the Monarch Platform could define what robotic-assisted diagnosis looks like for the next decade. That is the scale of the ambition, and the scale of the risk.
References
- Johnson & Johnson MedTech. (2025, October 28). Johnson & Johnson to Advance Robotics Development with NVIDIA Isaac for Healthcare. https://www.jnj.com/media-center/press-releases/johnson-johnson-to-advance-robotics-development-with-nvidia-isaac-for-healthcare
- Johnson & Johnson MedTech. (2025, March 12). Johnson & Johnson MedTech Announces Clearance of MONARCH QUEST for Enhanced Robotic-Assisted Bronchoscopy. https://www.jnj.com/media-center/press-releases/johnson-johnson-medtech-announces-clearance-of-monarchtm-quest-for-enhanced-robotic-assisted-bronchoscopy
- Johnson & Johnson. (2026, January 7). Johnson & Johnson Submits OTTAVA Robotic Surgical System to the U.S. Food and Drug Administration. https://www.jnj.com/media-center/press-releases/johnson-johnson-submits-ottava-robotic-surgical-system-to-the-u-s-food-and-drug-administration
- Reuter, E. (2025, October 28). J&J partners with Nvidia on AI for kidney stone procedures. MedTech Dive. https://www.medtechdive.com/news/jnj-nvidia-monarch-robot-urology/804057/
- Zipp, R. (2026, January 21). J&J’s cardiovascular business boosts medtech growth in 2025. MedTech Dive. https://www.medtechdive.com/news/jnj-q4-full-year-earnings-cardiovascular/810121/
- Kelly, S. (2019, May 21). J&J’s Auris unveils early results on Monarch robot for lung procedures. MedTech Dive. https://www.medtechdive.com/news/jjs-auris-unveils-early-results-on-monarch-robot-for-lung-procedures/555223/
- MedTech Dive. (2023, June 23). Intuitive’s Ion platform may provide “most exciting advancements” in robotic technology. https://www.medtechdive.com/news/intuitive-ISRG-JNJ-ion-platform-robot-surgery/653750/
- NVIDIA. (2026, March 16). NVIDIA and Global Robotics Leaders Take Physical AI to the Real World. NVIDIA Investor Relations. https://investor.nvidia.com/news/press-release-details/2026/NVIDIA-and-Global-Robotics-Leaders-Take-Physical-AI-to-the-Real-World/default.aspx
- Shah, K. (2026, February 16). J&J’s MedTech Shows Strong Growth in 2025: Will 2026 be Better? Yahoo Finance. https://finance.yahoo.com/news/j-js-medtech-shows-strong-145500319.html
- Surgical Robotics Technology. (2025, October 28). Johnson & Johnson Teams with NVIDIA Isaac for Healthcare to Advance Robotics Development. https://www.surgicalroboticstechnology.com/news/johnson-johnson-teams-with-nvidia-isaac-for-healthcare-to-advance-robotics-development/
- Comparing Monarch Versus Ion Robotic-Assisted Bronchoscopy Platforms. (2025). Journal of Bronchology and Interventional Pulmonology. https://pubmed.ncbi.nlm.nih.gov/40662912/
- Shape-Sensing Robotic-Assisted Bronchoscopy vs. Electromagnetic Navigation. (2026). Journal of Clinical Medicine. https://pmc.ncbi.nlm.nih.gov/articles/PMC12898066/
- Noah Medical. (2023). The Galaxy System Receives FDA Clearance for Robotic Navigated Bronchoscopy. https://www.noahmed.com/press-releases/the-galaxy-system-receives-fda-clearance-for-robotic-navigated-bronchoscopy
- Medtronic. (2026, February 17). Medtronic announces first surgery with Hugo robotic-assisted surgery system in the U.S. performed at Cleveland Clinic. https://news.medtronic.com/2026-02-17-Medtronic-announces-first-surgery-with-Hugo-TM-robotic-assisted-surgery-system-in-the-U-S-performed-at-Cleveland-Clinic
- CMR Surgical. (2025, December 16). CMR Surgical secures 510(k) clearance of its Versius Plus robotic surgical system. https://us.cmrsurgical.com/news/cmr-surgical-secures-510k-clearance-of-its-versius-plus-robotic-surgical-system
- MMR Statistics. (2026). Global Robotic Surgery Market Size (2025-2032). https://www.mmrstatistics.com/reports/795456/global-robotic-surgery-market